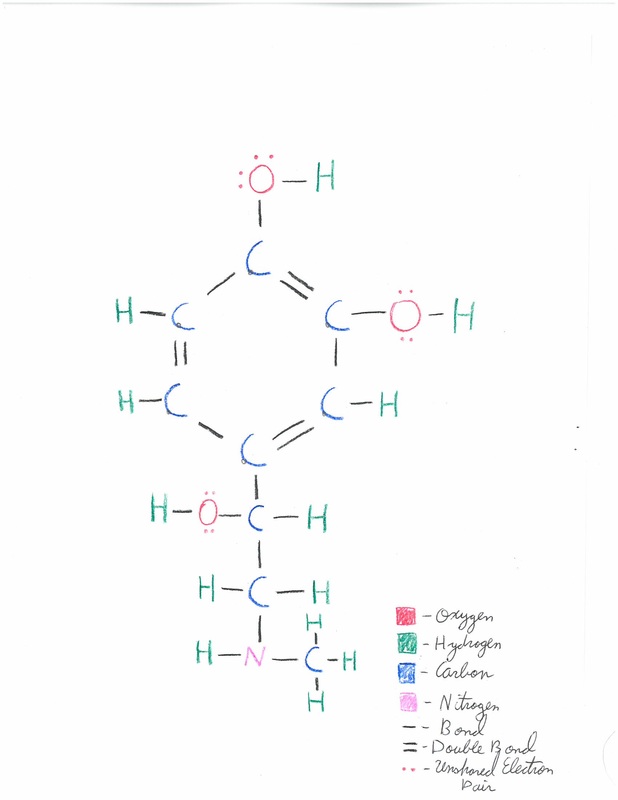
Bonding and VSEPR form:
An epinephrine molecule can be seen as composed of two distinct elements. Firstly, the six carbon benzene ring and it's attached molecules, which form a cyclical molecule. Secondly, the chain of carbon, hydrogen, and nitrogen which extends from the bottom of the benzene ring, which forms a linear molecule. Thus, the VSEPR form for epinephrine is linear and cyclical together.
Several types of bonds are present in epinephrine. Within the benzene ring, carbon bonds to carbon with absolutely covalent bonds. The bond between a carbon and an oxygen, as appears on the benzene rings "arms" is polar covalent. Oxygen has an electronegativity of 3.5, and carbons electronegativity value is 2.5 for a difference of 1. Oxygen also bonds to hydrogen with polar covalent bonds. Oxygen has an elecronegativity value of 3.5, and the electronegetivity value for hydrogen is 2.2. Thus, the difference between the values is 1.3. Carbon and nitrogen form strongly covalent bonds. Nitrogen has the electronegetivity value 3.0, so the difference between it's value and that of carbon, 3.5, is .5. Finally, nitrogen and hydrogen form moderately covalent bonds, with a difference in electronegativities of 1.8.